Keratoconus is a non-inflammatory eye condition in which the normally round dome-shaped cornea progressively thins causing a cone-like bulge to develop. This results in significant visual impairment.
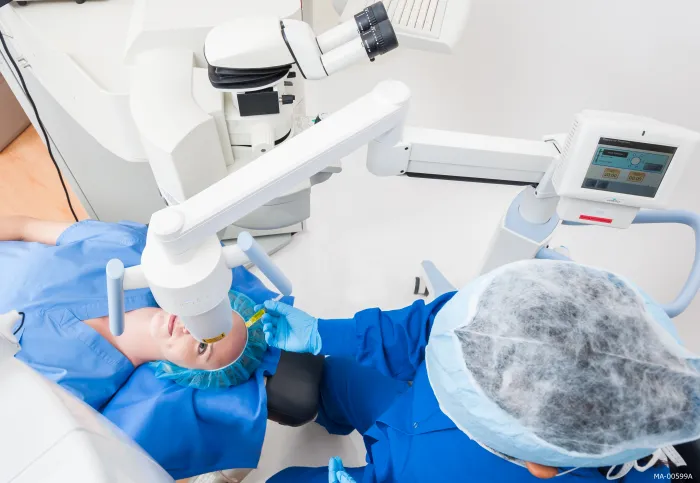
Corneal Cross-linking is a minimally invasive outpatient procedure for the treatment of progressive keratoconus. FDA-approved cross-linking combines the use of prescription eye drops, and ultra-violet A (UVA) light to create new collagen cross-links and leads to the stiffening of the cornea. The goal of the procedure is to stiffen the cornea to slow or prevent further progression of the condition and preserve your vision.